Gold nanorods (GNRs) are microscopic gold nanoparticles that are have been elongated to resemble rods, rather than the traditional spherical shape. Since the early 2000s, they have gotten much attention from researchers because of their tunable optical properties, which makes them suitable for a wide range of applications, from technological to medical.
Because of their shape and the fact they can be produced in different lengths, gold nanorods have a number of advantages over other gold nanoparticles. Their tunable optical properties make them ideal for use in diagnostic tests, while their improved absorption and scattering properties makes them potentially useful for drug delivery, cell imaging and photothermal therapy for cancer and liposuction.
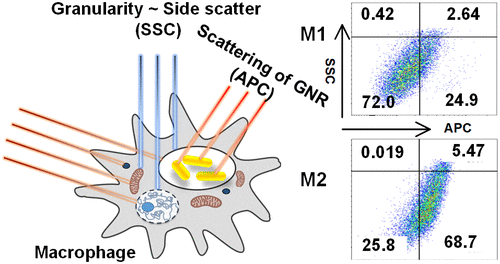
Now, researchers at Bar-Ilan University (BIU) in Ramat Gan (near Tel Aviv) have used gold nanorod scattering to identify the immune system’s killer and savior’ stages. Further development of this technology will lead to novel methods for predicting the development of diseases such as cancer, atherosclerosis, and fibrosis from simple tissue fluids or blood samples.
Every biological system is naturally equipped with a defense mechanism to protect against abnormal changes caused by either local, environmental, or biochemical alteration. White blood cells (WBC) play the role of such a ‘soldier’ in our immune response. One type of WBC known as macrophages is the most efficient and specialized fighter since it is simultaneously equipped with the power of selective identification and elimination of foreign invaders, as well as the powerto repair wounds.
Depending on their work distribution, macrophages are mainly comprised of two types, M1 and M2. M1 cells act as the ‘professional killer’, while M2 cells are more concentrated on healing activity. In a normal, healthy situation, the immune system maintains a good balance between M1 and M2 cells. But in diseased conditions like bacterial, virus or parasite infections or inflammations for atherosclerosis, cancer, or arthritis, the balance between M1 and M2 becomes affected, and depending on the crisis, a particular shift in M1 or M2 population occurs.
The shortcode is missing a valid Donation Form ID attribute.
If such changes could be monitored, it would lead to easy diagnostics and prediction of health conditions. There is currently no tool that can provide easy detection of M1/M2 cells directly from tissue fluid or a blood sample in a label-free manner without fluorescent tagging.
In the study just published in the journal Nano Letters by Dr. Ruchira Chakraborty – a leading researcher at Prof. Dror Fixler’s laboratory at BIU’s Faculty of Engineering and Institute of Nanotechnology and Advanced Materials – researchers from Bar-Ilan University in Israel have shown a simple solution to this issue with the help of the scattering effect of GNRs.
Gold-based nanoparticles are well known for their prominent optical property with high absorbance and scattering effects. By manipulating the scattering effect and adjusting the surface coating of GNRs, the researchers were able to identify changes in the optical property of M1 and M2 macrophages and utilize them as a parameter to monitor physiological changes.
“Our approach in utilizing the scattering of GNRs to identify M1 and M2 macrophages opens a new strategy in cellular identifications using FCM with the help of increased scattering of internalized nanoparticles,” said Chakraborty.
“Further development of this technique will lead us to build a new point of care or a biopsy tool which can predict the stages of manifestation of diseases like cancer, atherosclerosis, and fibrosis just from the simple tissue fluids or blood samples,” added Prof. Dror Fixler, director of BIU’s Nano Institute, who led the study in cooperation with Prof. Ran Kornowski and Dr. Dorit Leshem from the nearby Rabin Medical Center.



